By: Bradley Metz & David Tarpy
In the “girl-boss” dystopia that is the honey bee colony, drones are the dopey, do-nothings that are at best an annoyance, or at worst the signs of disorder and amplifier of parasites. Unlike the workers, queens, or even larvae, drones have no utility in the day-to-day operations of the honey bee colony. In fact, the boys have only a single, shamefully obvious task—to propagate their mother’s (usually the queen) genes to the next generation, flying en masse to mate with any virgin queen they can catch. This task is fatal to the drones, so even the fulfillment of this single job proves overwhelming. To make matters worse, honey bees are highly polyandrous, with several different drones mating with each queen. Therefore, the final fate of the honey bee male is to exist solely as a tiny proportion of stored spermatozoa to be meted out by the queen in the formation of future workers, themselves sterile dead ends, which busily contribute to colony growth, provisioning, and development. It is the rarest drone that will become a grandfather – siring a queen – and successfully pass his genes to the next generation.
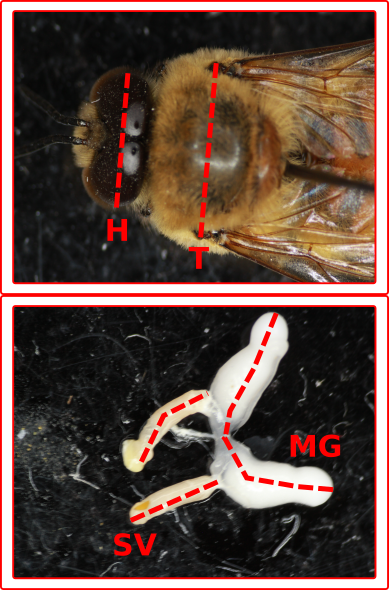
Figure 1: Measuring drones is conceptually
identical to how we measure queens. Body size measures are made using a microscope and a scale. We measure head and thorax width, and whole body and thoracic mass. Reproductive
potential is measured both with morphometrics: seminal vesicle and mucus gland length, and by measuring sperm count and viability using
fluorescent microscopy.
I was trained in beekeeping as part of my graduate work on chemical communication among larvae and adults. Drones were a nuisance; we pulled comb and froze the boys in their cells to minimize Varroa. Once, in a fit of bravado induced by an errant internet search, we fried the brood and served them at a party (they tasted like sweetened vegetable oil). So, when I started working for Dr. David Tarpy at NC State and the first project proposed was on the evaluation of drone reproductive health, my first thought was—“why?!” In my mind, these disposable social gametes were as unremarkable as a single spermatozoan, which (despite any Monty Python songs that might be evoked) are generally more interesting in aggregate than individually. Nevertheless, upon joining the Honey Bee Queen & Disease Clinic, I was tasked with doing for drones what Dr. Tarpy and colleagues had previously done for queens (see Tarpy et al., 2012 or my own prior article in this magazine): establish a measure of reproductive quality and variation to develop a national-level database so that we can begin to understand how and why drones vary, and the consequences of this for the queen and colony.
The first thing I had to get over was the idea that the production of drones was, in and of itself, pathological. In my myopic viewpoint, I neglected to consider that while yes, a colony that is anarchical or headed by an aging or poorly mated queen will produce an excess of drones, producing a seasonally appropriate number of drones is a sign of a colony doing well, growing sufficiently well so that it can commit to reproduction! Typically, a healthy colony will produce around 10% of their brood as drones, with that proportion increasing prior to swarming season (reviewed in Boes, 2010). However, colony size also plays a factor, as we learned (anew) that smaller colonies will neither rear nor foster drones. A colony that refuses to produce drones is likely not a colony that is in good shape. Drone production by healthy colonies plays into the second factor that I needed to understand, the overall ecology of the mating environment. Very few operations in the U.S. perform instrumental insemination at scale. That means that the million-plus queens produced commercially each year are largely mating on the wing, with little to no input from the breeders. That means whatever characteristics we want in the workers must be added indirectly by raising colonies for drone production (i.e., large, healthy colonies) that have desirable characteristics and that those drones are successful in mating. But which drones are successful?! Also, what are the characteristics of a successful drone for the traits we want in a daughter colony? That’s a thornier question.
For a drone to successfully mate, they typically need to be LARGE. The research on this usually compares drones reared in drone cells to those reared in worker cells to create a bimodal (two humped) size distribution to show that small drones produce less sperm, are less likely to successfully mate, and tend to sire a lower-than-expected proportion of the worker population (reviewed in Gençer and Kahya, 2020). So bigger is better, right? We-ell, not necessarily, since smaller workers appear to exhibit differential flight behaviors consistent with different mating strategies or life histories (see Couvillon et al., 2010), suggesting the little guys might have other plans. Also, because siring workers is a genetic dead end, the real prize is siring a queen, and it’s not necessarily the father of the most workers that succeeds there. In fact, rather than nepotistically rear their own super sisters, workers tend to select larvae from relatively rare patrilines to become queens (see Withrow & Tarpy 2018 for a recent example). Therefore, maybe size isn’t the only metric important for drones. Certainly, we know from sampling throughout the country that drones adapted to the climate in the southwest tend to be smaller, although no less fecund, than their more northern brethren and they seem to do just fine. That then leaves the other main characteristic of import for a drone: their sperm.
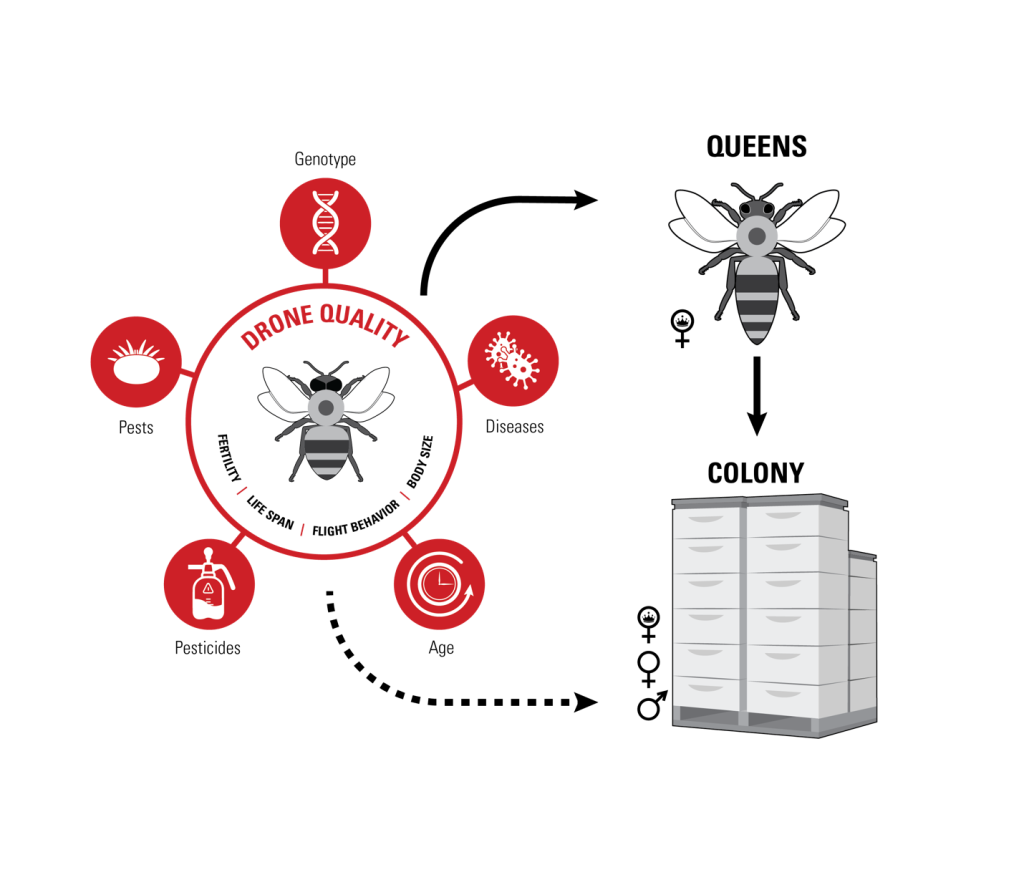
Figure 2: Impact of drones. Measuring drones and the impact of modern mechanized agriculture on their fertility is important for its own end, and to understand potential threats to male reproductive in general. However, the more proximal importance is for understanding how drone quality impacts queen through sperm with lower longevity or other subtle problems.
Since his sperm will far outlive the adult drone, sperm viability at ejaculation and their longevity in the queen are the prime traits which we study. Knowing that a queen spends a ridiculously meager two spermatozoa per egg, lays something like 2,000 eggs a day at peak, we know that to keep the queen laying for the year, we’d want something like 1.5 million sperm (numbers from Baer et al., 2016). Since the average queen from our Queen Clinic database stores about five million sperm, that should give a queen a theoretical lifespan of around four years, if sperm count were the only determinant in her longevity. However, only about 85% of those sperm are viable on average, and since we largely measure queens shortly after production, that’s a big hit to a queen’s stores right off the bat. Thus, it’s incumbent to understand the factors that affect sperm during storage so that this number doesn’t drop even lower.
Even with the best intentions, I still tend to only think about drones in the context of the wider superorganism. But how important are the drones to the colony? It’s unclear how much the health of the drone impacts the health of the stored sperm –which is what we really care about. It’s commonly thought that only live sperm will make it into the spermatheca, so the quality of the drone shouldn’t matter. Although there’s a paucity of data on this, an interesting study by Kairo et al., (2016) showed that drones who were exposed to fipronil exhibited signs of reduced sperm quality but also passed those declines on to the queen. This means that poisoned drones probably provide sperm with a lower shelf life! The laundry list of things that can impact drone reproductive potential is huge (reviewed most recently in Rangel and Fisher, 2019) and all are familiar to anyone in beekeeping today (e.g. parasites, pesticides, temperature fluctuation and disease). The challenge now is to more firmly draw the line from effects on drones to effects on the queen and colony. The prevailing theory I’m therefore operating under, while building this database of drone quality, is that drones can be a prior-generation red flag—the rose bush in the vineyard that will enable us to detect potential problems with reproduction before they have a chance to impact the next generation’s queens. The next steps involve testing drones stressed in various ways and following those drones through the mating process into the next generation.
I want to learn more about drones. The nation-wide database we’ve built at the Clinic has set a baseline for the expected size, reproductive potential and the relationship between the two. We must now learn what disrupts that relationship, what impact that has on colony health and reproduction and what levers we possess to manipulate it. I wish I’d have an answer, but I only have a long list of questions. One of the greatest aspects of the Clinic from my perspective is that I’m not asking questions in an ivory tower; when I interface with beekeepers, it’s their ideas, questions, and perspectives I develop. I then explore those questions until we can learn more about what these little boys are made of after all.
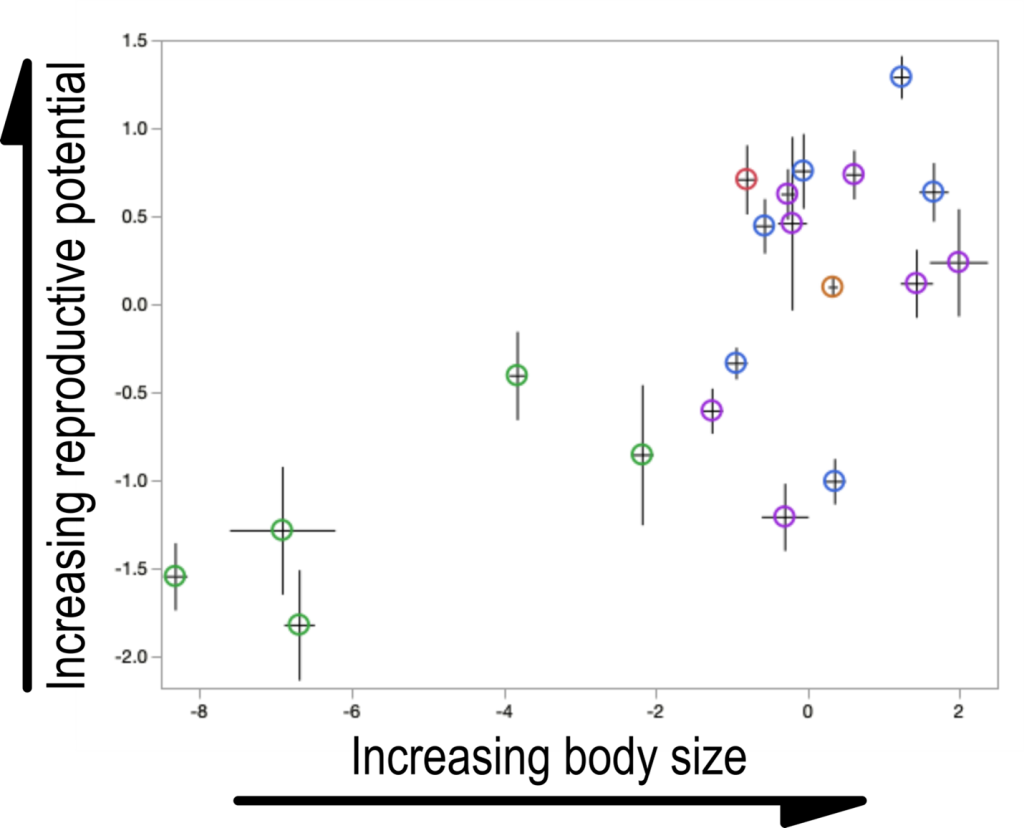
Figure 3: Colony variation in drones. Colonies vary significantly in the size and reproductive quality of the drones they produce. While this has well-established impacts on the competitive abilities of drones, it’s unclear whether smaller, less fertile drones represent a pathological colony condition, differential reproductive choice, or simply random variation. Though we tend to think larger drones means better drones, the best advice we can give the beekeeper is to ensure that there are plenty
of drones flying and trust the natural mating process to sort things out. These data are reported on more fully in Metz & Tarpy, 2021.
Acknowledgments: This work in progress was funded by the California State Beekeepers Association, the USDA, and is supported by beekeeper clients of the NC State Queen & Disease Clinic.
Contact Bradley Metz at bnmetz@ncsu.edu
References
Baer, B., Collins, J., Maalaps, K., & den Boer, S. P. A. (2016). Sperm use economy of honeybee (Apis mellifera) queens. Ecology and Evolution, 6(9), 2877–2885. https://doi.org/10.1002/ece3.2075
Boes, K. E. (2010). Honey bee colony drone production and maintenance in accordance with environmental factors: an interplay of queen and worker decisions. Insectes Sociaux, 57(1), 1–9. https://doi.org/10.1007/s00040-009-0046-9
Couvillon, M. J., Hughes, W. O. H., Perez-Sato, J. A., Martin, S. J., Roy, G. G. F., & Ratnieks, F. L. W. (2010). Sexual selection in honey bees: colony variation and the importance of size in male mating success. Behavioral Ecology, 21(3), 520–525. https://doi.org/10.1093/beheco/arq016
Kairo, G., Provost, B., Tchamitchian, S., Abdelkader, F. ben, Bonnet, M., Cousin, M., Sénéchal, J., Benet, P., Kretzschmar, A., Belzunces, L. P., & Brunet, J.-L. (2016). Drone exposure to the systemic insecticide Fipronil indirectly impairs queen reproductive potential. Scientific Reports, 6(31904). https://doi.org/10.1038/srep31904
Rangel, J., & Fisher, A. (2019). Factors affecting the reproductive health of honey bee (Apis mellifera) drones—a review. Apidologie, 1–20. https://doi.org/10.1007/s13592-019-00684-x
Metz, B. N., and D. R. Tarpy. (2021). Reproductive and Morphological Quality of Commercial Honey Bee (Hymenoptera: Apidae) Drones in the United States. J. Insect Sci. 21: 2–3. https://doi.org/10.1093/jisesa/ieab048
Gençer, H. V., & Kahya, Y. (2020). Sperm competition in honey bees (Apis mellifera L.): the role of body size dimorphism in drones. Apidologie, 51, 1–17. https://doi.org/10.1007/s13592-019-00699-4
Tarpy, D. R., Keller, J. J., Caren, J. R., & Delaney, D. A. (2012). Assessing the mating “health” of commercial honey bee queens. J. Econ. Entomol, 105(1), 20–25. https://doi.org/10.1603/EC11276
Withrow, J. M., & Tarpy, D. R. (2018). Cryptic “royal” subfamilies in honey bee (Apis mellifera) colonies. PLoS ONE, 13(7), 1–11. https://doi.org/10.1371/journal.pone.0199124








